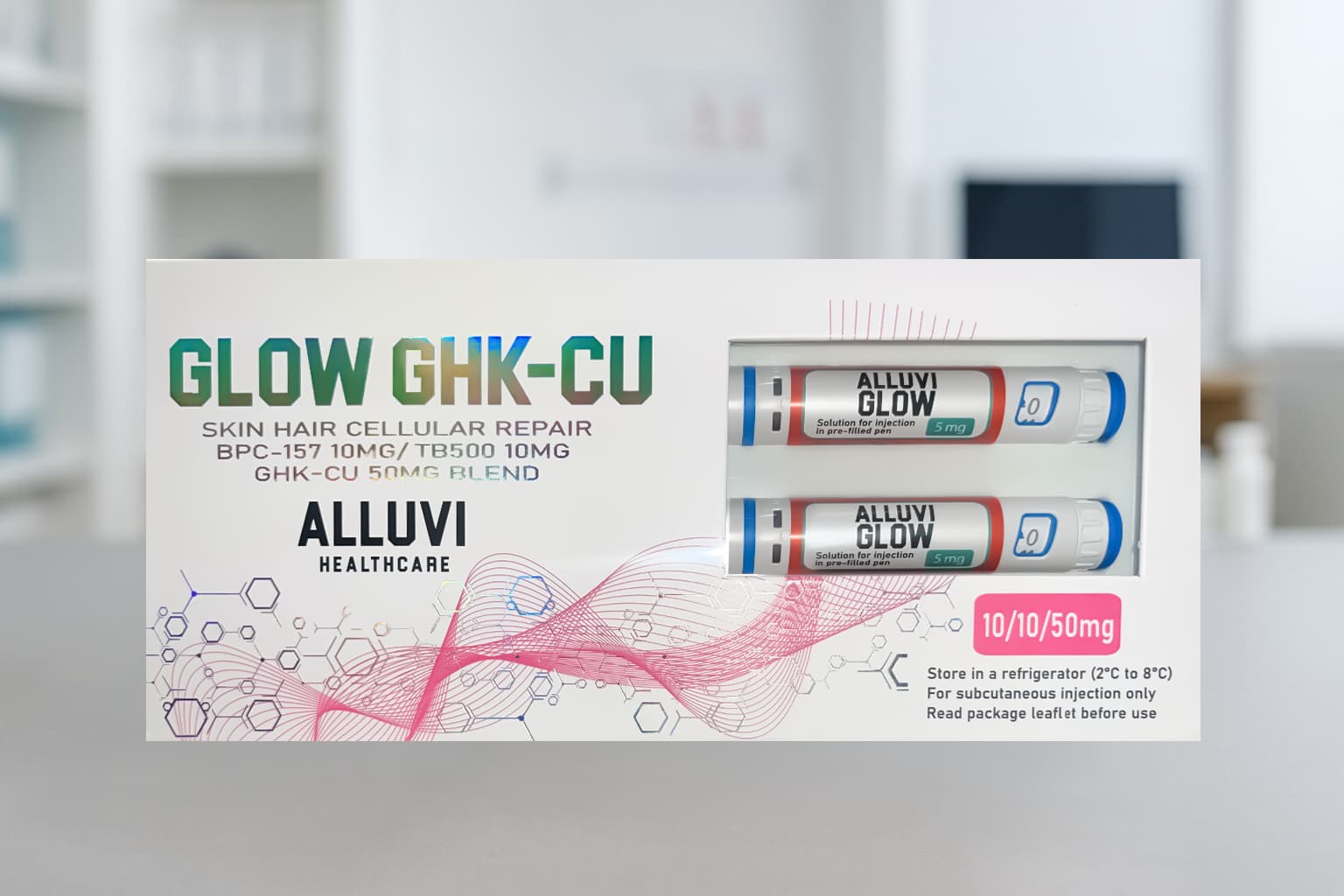
What Is the Alluvi Glow 70mg Pen?
The Alluvi Glow 70mg Pen is a Synedica-certified, pre-filled tri-peptide research injection pen delivering 70mg of three of the most powerful skin, hair and cellular repair peptides in modern research science — BPC-157 (10mg), TB-500 (10mg) and GHK-Cu (50mg) — combined in a single precision-engineered pre-filled pen. Supplied exclusively by Alluvi Healthcare as part of the Synedica-certified research pen range, the Alluvi Glow 70mg pen is the most comprehensive skin cellular repair, hair regeneration and dermatological research tool available from a UK domestic source.
GHK-Cu — Copper Peptide GHK-Cu — is one of the most extensively studied bioactive tripeptides in dermatological and regenerative biology, with over 40 years of published research demonstrating potent wound healing, collagen synthesis stimulation, antioxidant activity, anti-inflammatory effects and hair follicle regeneration. Combined with BPC-157’s tissue repair and angiogenesis mechanisms and TB-500’s actin-regulating cell migration properties, the Alluvi Glow 70mg pen addresses the complete spectrum of skin and hair cellular repair biology from three distinct and complementary peptide pathways. UK researchers searching for the most verified, comprehensively formulated GHK-Cu research pen UK will find everything they need at Alluvi Healthcare.
The Science — BPC-157 + TB-500 + GHK-Cu Tri-Peptide Mechanism
The scientific rationale for combining BPC-157, TB-500 and GHK-Cu in a single research pen lies in their deeply complementary mechanisms across the skin and hair repair cascade. Each peptide addresses a distinct biological pathway — yet all three converge on the same research outcome: comprehensive skin cellular repair, hair follicle regeneration and dermal tissue restoration. Together in the Alluvi Glow 70mg pen, they represent the most mechanistically complete skin and hair research tool available in the UK today.
Key Research Mechanisms — GHK-Cu (Copper Peptide)
- Collagen and elastin synthesis: GHK-Cu stimulates collagen I, III and IV synthesis and elastin production — fundamental to skin structural integrity and wound healing research
- Wound healing acceleration: GHK-Cu has demonstrated accelerated wound contraction, epithelialisation and dermal repair in over 40 years of published pre-clinical research
- Hair follicle regeneration: GHK-Cu stimulates hair follicle enlargement and hair growth in pre-clinical models — one of the most cited applications in GHK-Cu research literature
- Antioxidant and anti-inflammatory: GHK-Cu activates superoxide dismutase and catalase, significantly reducing oxidative stress and inflammatory cytokine expression in skin tissue models
- Gene expression regulation: GHK-Cu modulates over 4,000 genes in human fibroblasts — upregulating repair and regeneration pathways and downregulating inflammatory and cancer-related gene expression
- Angiogenesis: GHK-Cu promotes neovascularisation in wound healing models — complementing BPC-157 and TB-500 VEGF-mediated angiogenesis mechanisms
- Skin barrier restoration: GHK-Cu upregulates genes involved in epidermal barrier function — relevant to atopic dermatitis, psoriasis and skin barrier dysfunction research
Key Research Mechanisms — BPC-157 (10mg)
- Tissue repair and angiogenesis: BPC-157 upregulates VEGFR2 expression and promotes neovascularisation critical for wound healing and skin repair research
- Anti-inflammatory: BPC-157 modulates NO signalling and reduces inflammatory cytokine expression — addressing the inflammatory phase of the wound repair cascade
- Fibroblast activation: BPC-157 activates dermal fibroblasts and promotes their migration into wound beds — accelerating the proliferative phase of skin repair
Key Research Mechanisms — TB-500 (10mg)
- Cell migration: TB-500 sequesters G-actin, enabling keratinocyte and fibroblast migration into wound sites — the critical first step in dermal wound closure
- Hair follicle stem cell activation: TB-500 activates hair follicle stem cells through Wnt pathway modulation — one of the most significant hair regeneration mechanisms in current research
- Wound closure acceleration: TB-500 has demonstrated accelerated dermal wound closure through keratinocyte migration in multiple pre-clinical models
Full Technical Specifications — Alluvi Glow 70mg
| Product Name | Alluvi Glow 70mg (Synedica) |
| Manufacturer | Synedica (distributed by Alluvi Healthcare UK) |
| Active Compounds | GHK-Cu 50mg + BPC-157 10mg + TB-500 10mg |
| Compound Class | Tri-Peptide · Skin Cellular Repair · Hair Regeneration · Wound Healing |
| Total Per Order | 70mg (2 pens × 35mg) |
| Dose Structure | 2 pens · each pen delivers GHK-Cu 25mg + BPC-157 5mg + TB-500 5mg × 2 doses |
| Delivery Format | Pre-filled subcutaneous injection pen |
| Needles Included | 6 × fine-gauge subcutaneous pen needles |
| Purity Standard | ≥98% — independent third-party analytical verification per compound |
| Certificate of Analysis | Batch-specific COA included with every order |
| Certification | Synedica — pharmaceutical-grade manufacturing standard |
| Storage — Unopened | Refrigerated 2–8°C · Away from direct light · Do not freeze |
| Storage — In Use | Refrigerate between uses · Use within stated stability window |
| Dispatch Method | Cold-chain insulated packaging · UK domestic stock |
| Dispatch Timeframe | Within 48 hours of order confirmation |
| Delivery Coverage | All UK addresses — mainland + highlands |
| Research Application | Skin cellular repair · Hair regeneration · Wound healing · Collagen synthesis · Dermatology |
| Regulatory Status | Research use only — not MHRA licensed for clinical use |
Alluvi Glow 70mg vs Single-Peptide Skin Research
| Feature | Alluvi Glow 70mg | GHK-Cu Only | BPC-157 Only |
|---|---|---|---|
| Peptide Coverage | GHK-Cu + BPC-157 + TB-500 (triple) | GHK-Cu only | BPC-157 only |
| Collagen Synthesis | ✓ GHK-Cu dominant | ✓ Yes | Limited |
| Hair Regeneration | ✓ GHK-Cu + TB-500 | ✓ Partial | ✗ No |
| Wound Healing | ✓ All 3 pathways | Partial | Partial |
| Angiogenesis | ✓ Triple mechanism | ✓ Limited | ✓ VEGFR2 |
| Anti-inflammatory | ✓ All 3 pathways | ✓ Antioxidant | ✓ NO system |
| Synedica Certified | ✓ Yes | Varies | Varies |
| COA ≥98% | ✓ Yes | Varies | Varies |
| 48hr UK Dispatch | ✓ Yes | Varies | Varies |
| Order | Buy Now → | — | — |
Research Applications — What UK Laboratories Are Investigating
- Wound healing biology: Investigating the complete wound repair cascade — haemostasis, inflammation, proliferation and remodelling — using the tri-peptide GHK-Cu + BPC-157 + TB-500 model
- Hair follicle regeneration: GHK-Cu and TB-500 synergistic effects on hair follicle stem cell activation, follicle enlargement and hair growth — a major area of UK dermatology and cosmetic research
- Collagen and extracellular matrix research: GHK-Cu stimulation of collagen I, III, IV and elastin synthesis in dermal fibroblast models
- Skin ageing biology: Investigating GHK-Cu-mediated gene expression changes and anti-ageing mechanisms in aged skin tissue models — relevant to longevity and cosmetic biology research
- Atopic dermatitis and psoriasis models: GHK-Cu skin barrier restoration and anti-inflammatory effects in inflammatory dermatosis research models
- Scar tissue and fibrosis research: GHK-Cu and BPC-157 anti-fibrotic effects in hypertrophic scar and keloid tissue models
- Oxidative stress and antioxidant research: GHK-Cu superoxide dismutase and catalase activation in skin oxidative stress models
- Combination protocols: Widely paired with Alluvi BPC-157 TB500 40mg and Alluvi NAD+ 1000mg in comprehensive multi-compound longevity and repair research programmes
Why Choose Alluvi Healthcare for GHK-Cu Research UK
UK researchers searching for GHK-Cu research pen UK, Alluvi Glow, or the most verified tri-peptide skin research pen consistently choose Alluvi Healthcare. As Britain’s most trusted Synedica-certified research compound pen supplier, Alluvi Healthcare delivers Synedica certification, ≥98% independently verified purity, pre-filled pen format with no reconstitution required, 48hr UK cold-chain dispatch, WhatsApp research support on +44 7537 823566, and the full range from a single UK source.
How to Order — Alluvi Glow 70mg
- Visit alluviukshop.co.uk/shop and select the Alluvi Glow 70mg Pen
- Consider adding the Alluvi BPC-157 TB500 40mg, Alluvi NAD+ 1000mg or Alluvi Retatrutide 40mg Pen for a comprehensive multi-compound research protocol
- Apply code alluvicare26 at checkout for 35% off — applied automatically on first orders
- Receive within 48 hours — cold-chain dispatched in discreet unmarked packaging with full tracking
- Refrigerate immediately at 2–8°C and verify the batch COA before beginning your research protocol
Storage & Handling
- Refrigerate immediately on receipt at 2–8°C
- Do not freeze — freezing permanently degrades peptide structure
- Protect from direct light — store in original packaging inside the refrigerator
- Do not shake or agitate — handle gently at all times
- Verify COA batch number before beginning any research protocol
- Note the expiry date printed on the pen body and outer packaging
- Inspect before use — if cloudy, discoloured or contains particulates do not use. Contact Alluvi Healthcare via WhatsApp immediately
Explore the Complete Alluvi Healthcare Research Range
- Alluvi Retatrutide 40mg Pen — Triple GLP-1/GIP/Glucagon · Synedica · 4×10mg · ≥98% COA
- Alluvi Retatrutide 20mg Pen — Initiation phase triple agonist · Synedica · 2×10mg
- Alluvi Tirzepatide 20mg Pen — Dual GLP-1/GIP agonist · Synedica · 2×10mg · ≥98% COA
- Alluvi BPC-157 TB500 40mg — Recovery and repair · Dual peptide · 4×10mg · ≥98% COA
- Alluvi NAD+ 1000mg — Brain cellular repair · Anti-ageing · 2×500mg · ≥98% COA
- Wholesale Orders — Bulk supply for UK laboratories
- Alluvi Healthcare Blog — Research insights, reviews and sourcing guides
⚠️ Research Use Only — Legal Disclaimer: The Alluvi Glow 70mg Pen is supplied exclusively for legitimate scientific laboratory research, analytical testing and formulation development. Not licensed as a medicine by the MHRA or any other regulatory body. Not for human consumption, self-administration, veterinary use or clinical application outside of authorised clinical trials. By purchasing, the buyer confirms they are a qualified researcher aged 18 or above operating in full compliance with all applicable UK regulations. For regulatory guidance visit MHRA.gov.uk. For clinical context visit NICE.org.uk.

Reviews
There are no reviews yet.