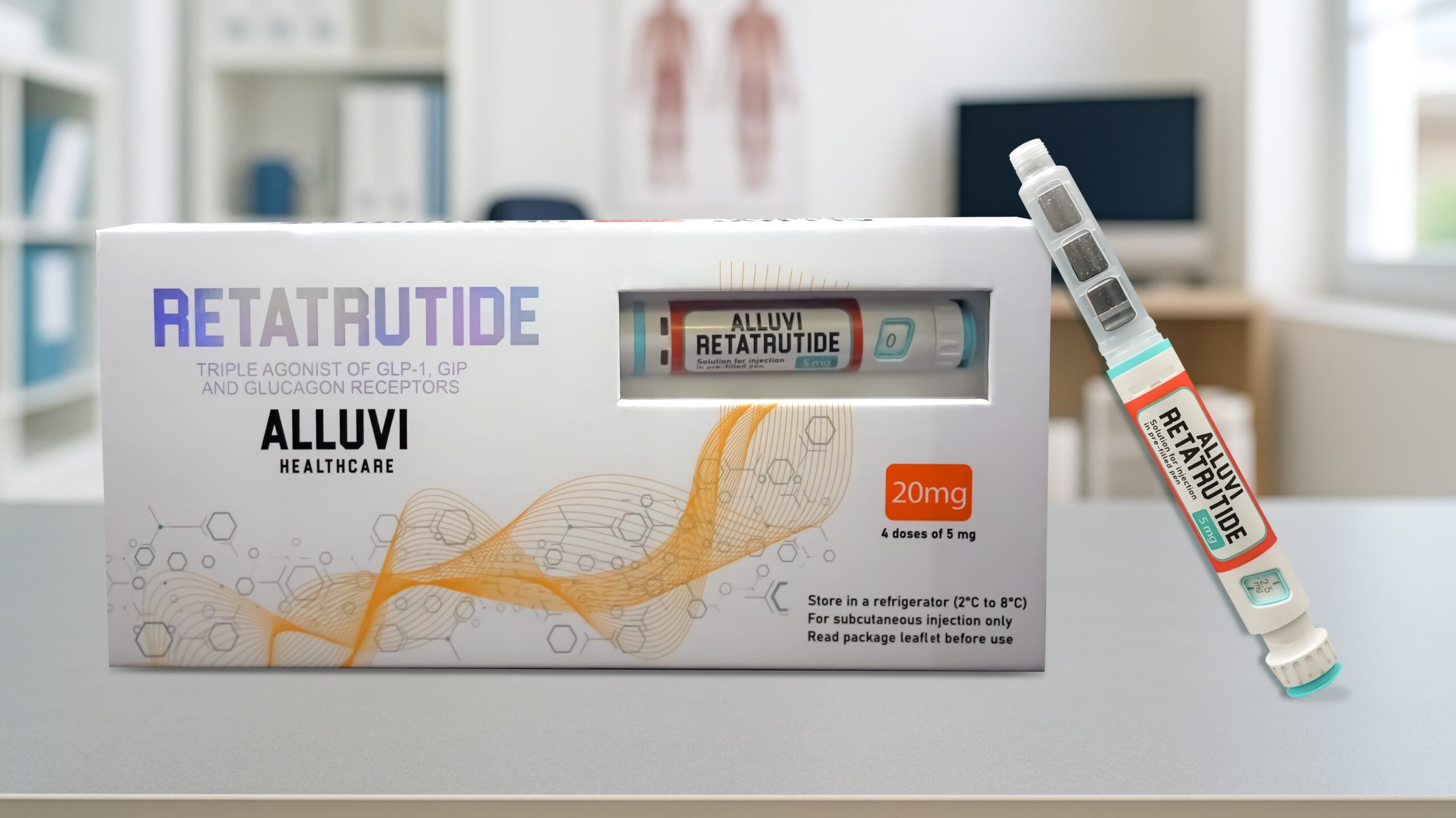
What Is Retatrutide 20mg (R&D Only)?
Retatrutide 20mg (R&D Only) is a research-grade triple GLP-1/GIP/Glucagon receptor agonist peptide compound supplied by Alluvi Healthcare exclusively for legitimate scientific research, analytical testing and formulation development. Presented in a Synedica-certified, pre-filled subcutaneous injection pen delivering 20mg of research-grade retatrutide across 2×10mg precision click-dial doses, this is the initiation-phase R&D format designed for UK laboratories beginning systematic retatrutide research programmes, establishing dose-response baselines, or running dual vs triple agonist comparative study designs.
As an R&D-only compound, Retatrutide 20mg from Alluvi Healthcare is supplied with full Synedica pharmaceutical-grade manufacturing certification, independent third-party COA verification at ≥98% purity, and all the cold-chain dispatch integrity that serious UK research demands. For UK researchers looking to buy retatrutide UK for laboratory use, Alluvi Healthcare is Britain’s most trusted domestic R&D source — delivering verified research-grade retatrutide pens within 48 hours from UK domestic stock.
The Science — Retatrutide Triple-Agonist R&D Mechanism
Retatrutide’s triple-receptor architecture — simultaneously targeting GLP-1, GIP and Glucagon receptors in a single engineered peptide molecule — produces the broadest and most potent metabolic R&D profile of any GLP-1-based compound available for UK laboratory research today. The GLP-1 component stimulates glucose-dependent insulin secretion, suppresses glucagon release and activates CNS appetite regulation pathways. The GIP component potentiates insulin secretion through a distinct receptor pathway and promotes adipose tissue lipid metabolism. The Glucagon component — the critical mechanistic differentiator from dual-agonist compounds — stimulates hepatic glucose production modulation, drives lipolysis and dramatically increases energy expenditure. Together these three receptor pathways produce synergistic metabolic effects that no single or dual agonist compound can replicate.
The landmark Phase 2 retatrutide trial published in the New England Journal of Medicine demonstrated dose-dependent weight reductions of up to 24.2% at 48 weeks — the largest pharmacological weight reduction ever recorded in a clinical trial at the time of publication. For UK R&D laboratories, Retatrutide 20mg (R&D Only) from Alluvi Healthcare provides the validated initiation-phase triple-agonist compound with the purity, certification and UK-domestic supply reliability that serious research demands. Full reference literature available on PubMed and ClinicalTrials.gov.
Key R&D Research Mechanisms — Retatrutide 20mg
- Triple receptor agonism at initiation dose: 2×10mg doses establishing the full triple GLP-1/GIP/Glucagon agonist baseline for systematic R&D protocol development
- Glucagon receptor R&D: The critical mechanistic differentiator from dual agonists — enabling UK researchers to investigate glucagon receptor contribution to metabolic outcomes at initiation-phase concentrations
- Hepatic fat oxidation research: Glucagon-driven hepatic fat oxidation and glucose production modulation — measurable from the 20mg initiation dose and directly comparable to dual-agonist controls
- Dose-response R&D design: The 20mg format is the standard initiation point for dose-escalation research designs building toward the Alluvi Retatrutide 40mg Pen sustained phase
- Comparative dual vs triple agonist R&D: Pairing retatrutide 20mg with Alluvi Tirzepatide 20mg enables the most mechanistically informative dual vs triple agonist comparative R&D design available in UK pre-clinical science
- Adipose tissue R&D: Triple-receptor synergistic lipolysis, white-to-beige adipose transition and fat oxidation investigations at initiation doses
- Metabolic syndrome R&D: Investigating the combined GLP-1/GIP/Glucagon mechanism in insulin resistance, dyslipidaemia and obesity-related metabolic dysfunction research models
Full Technical Specifications — Retatrutide 20mg (R&D Only)
| Product Name | Retatrutide 20mg (R&D Only) — Alluvi Healthcare |
| Supplier | Alluvi Healthcare UK (Synedica certified) |
| Active Compound | Retatrutide |
| Compound Class | Triple GLP-1 / GIP / Glucagon Receptor Agonist |
| Research Classification | R&D Only — Research & Development use exclusively |
| Total Per Pen | 20mg |
| Dose Structure | 2 × 10mg doses (precision click-dial mechanism) |
| Micro-Dose Increments | 2.5mg · 5mg · 7.5mg · 10mg per dose |
| Delivery Format | Pre-filled subcutaneous injection pen |
| Needles Included | 6 × fine-gauge subcutaneous pen needles |
| Purity Standard | ≥98% — independent third-party analytical verification |
| Certificate of Analysis | Batch-specific COA included with every order |
| Certification | Synedica — pharmaceutical-grade manufacturing standard |
| Storage — Unopened | Refrigerated 2–8°C · Away from direct light · Do not freeze |
| Storage — In Use | Refrigerate between administrations · Use within stability window |
| Dispatch Method | Cold-chain insulated packaging · UK domestic stock |
| Dispatch Timeframe | Within 48 hours of order confirmation |
| Delivery Coverage | All UK addresses — mainland + highlands |
| Research Application | Initiation-phase triple GLP-1/GIP/Glucagon R&D · Dose-response · Comparative metabolic research |
| Regulatory Status | R&D Only — not MHRA licensed for clinical use |
Retatrutide 20mg R&D vs Other Formats — Choosing the Right Research Compound
| Feature | Retatrutide 20mg (R&D Only) | Retatrutide 40mg | Tirzepatide 20mg |
|---|---|---|---|
| Receptor targets | GLP-1 + GIP + Glucagon | GLP-1 + GIP + Glucagon | GLP-1 + GIP only |
| Total per pen | 20mg | 40mg | 20mg |
| Doses per pen | 2 × 10mg | 4 × 10mg | 2 × 10mg |
| R&D classification | R&D Only | R&D Only | R&D Only |
| Protocol position | Initiation phase baseline | Sustained phase endpoint | Dual-agonist comparator |
| Synedica certified | ✓ Yes | ✓ Yes | ✓ Yes |
| COA ≥98% | ✓ Yes | ✓ Yes | ✓ Yes |
| Cold-chain 48hr UK | ✓ Yes | ✓ Yes | ✓ Yes |
| Order | Buy → | Buy → | Buy → |
R&D Applications — What UK Laboratories Are Investigating
- Initiation-phase triple-agonist R&D: Establishing the baseline triple GLP-1/GIP/Glucagon agonist research profile at 2×10mg before protocol escalation
- Dose-response R&D: Investigating dose-dependent pharmacological outcomes across initiation (20mg) and sustained (40mg) formats in controlled laboratory conditions
- Dual vs triple agonist comparative R&D: Using retatrutide 20mg alongside tirzepatide 20mg to isolate the specific glucagon receptor contribution to metabolic research outcomes
- Hepatic steatosis R&D: Glucagon-mediated hepatic fat oxidation and NAFLD/NASH-relevant pathway research from the initiation dose
- Weight loss mechanism R&D: Investigating the triple-agonist weight reduction cascade referenced to the landmark NEJM Phase 2 retatrutide data
- Adipose biology R&D: GIP + Glucagon synergistic effects on adipose lipolysis, white-to-beige transition and fat oxidation at initiation-phase concentrations
- Compound pharmacy retatrutide R&D: Baseline compound characterisation, stability analysis and formulation development research using verified Synedica-manufactured retatrutide
- Multi-compound R&D programmes: Widely paired with Alluvi NAD+ 1000mg, Alluvi BPC-157 TB500 40mg and Alluvi Glow 70mg
Where to Buy Retatrutide UK — Why Alluvi Healthcare
UK researchers searching for “where to buy retatrutide UK”, “retatrutide uk buy online uk”, “reta uk”, “retatrutide uk source”, “retatrutide pens uk” or “buy retatrutide uk” consistently arrive at Alluvi Healthcare — and for good reason. As Britain’s most trusted Synedica-certified R&D compound pen supplier, Alluvi Healthcare is the definitive answer to the question of where to buy retatrutide UK. Every pen is Synedica certified, independently COA verified at ≥98%, pre-filled for research precision, and cold-chain dispatched within 48 hours from UK domestic stock with no import delays, no customs risk and no temperature excursion uncertainty.
How to Order — Retatrutide 20mg (R&D Only)
- Visit alluviukshop.co.uk/shop and select Retatrutide 20mg (R&D Only)
- Consider adding the Retatrutide 40mg (R&D Only) for a complete initiation-to-sustained R&D protocol, or Tirzepatide 20mg for a dual vs triple comparative design
- Apply code alluvicare26 at checkout for 35% off — applied automatically on first orders
- Receive within 48 hours — cold-chain dispatched in discreet unmarked packaging with full tracking
- Refrigerate immediately at 2–8°C and verify the batch COA before beginning your R&D protocol
Storage & Handling — R&D Grade Requirements
- Refrigerate immediately on receipt at 2–8°C
- Do not freeze — freezing permanently degrades retatrutide’s molecular structure and invalidates R&D results
- Protect from direct light — store in original packaging inside the refrigerator
- Do not shake or agitate — handle gently at all times to prevent peptide aggregation
- Verify COA batch number before beginning any R&D protocol administration
- Note the expiry date printed on pen body and outer packaging
- Inspect before use — if cloudy, discoloured or contains particulates do not use. Contact Alluvi Healthcare via WhatsApp immediately
Explore the Complete Alluvi Healthcare R&D Research Range
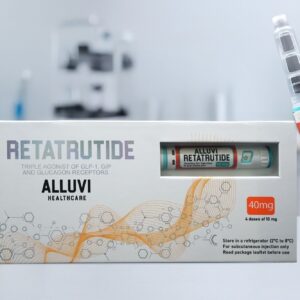
- Retatrutide 40mg (R&D Only) — Sustained phase · Triple agonist · 4×10mg · ≥98% COA · 48hr UK dispatch
- Tirzepatide 40mg (R&D Only) — Sustained dual GLP-1/GIP · 4×10mg · Synedica · ≥98% COA
- Tirzepatide 20mg (R&D Only) — Initiation dual GLP-1/GIP · 2×10mg · Synedica · ≥98% COA
- BPC-157 TB500 40mg (R&D Only) — Recovery and repair · Dual peptide · 4×10mg · ≥98% COA
- Glow 70mg (R&D Only) — GHK-Cu + BPC-157 + TB-500 · Skin · hair · cellular repair
- NAD+ 1000mg (R&D Only) — Brain cellular repair · Anti-ageing · 2×500mg · ≥98% COA
- Wholesale R&D Supply — Bulk supply for UK research institutions and laboratories
- Alluvi Healthcare Blog — Where to buy retatrutide UK · Reviews · Sourcing guides · R&D insights
⚠️ R&D Only — Legal Disclaimer: Retatrutide 20mg (R&D Only) is supplied by Alluvi Healthcare exclusively for legitimate scientific laboratory research, analytical testing and formulation development. Not licensed as a medicine by the MHRA or any other regulatory body. Not for human consumption, self-administration, veterinary use or clinical application outside of authorised clinical trials. By purchasing, the buyer confirms they are a qualified researcher aged 18 or above operating within a legitimate research context in full compliance with all applicable UK regulations, institutional governance frameworks and ethical guidelines. Alluvi Healthcare accepts no liability for misuse outside of these stated conditions. For regulatory guidance visit MHRA.gov.uk. For cl
Reviews
There are no reviews yet.